Objectives
Students will know how temperature affects dissolved oxygen and be able to create a graph showing this relationship.
Overview
Rating:
1. Students think about the adaptations of animals to live in different water temperatures
2. Students test four different water temperatures for dissolved oxygen
3. Students report and discuss the implications of increased temperature on dissolved oxygen
4. Students use Hudson River data to look at the impact of temperature and dissolved oxygen throughout the seasons
Materials
For demonstration:
- Cups with hot and cold water
- Ice
For each group:
- Copies of lab sheet
- Dissolved oxygen test kit
- Thermometer (up to 100ºC)
- One jar with water at room temperature
- One jar with water and ice
- One jar with water that was boiled for 20 minutes and then cooled (sealed)
- One jar with water that was heated to 50º C for several hours and then cooled (sealed)
Procedure
Preparation: Prepare enough jars of water so that each group will have one at room temperature, one with ice, and one of each boiled sample. Leave the ice water jars open to the air, adding ice to cool them to 5ºC (they can be left open in a refrigerator or ice chest). The jars at room temperature should be left open at least overnight, since some schools’ water tanks have very low levels of dissolved oxygen. Prepare two canned samples: one set of samples should be water that was heated to 50º C and held at that temperature for several hours, then poured carefully into jars and sealed (a coffee maker is an easy way to do this; just fill up with water and leave heated for several hours, then pour into canning jars and seal). The other set of heated samples should be brought to boiling for 20 minutes and then poured into jars and sealed. This preparation can be done anytime ahead of class; the jars will be cooled when the students use them, but the amount of DO inside will have been ‘fixed’. You can even do this several weeks ahead of time. Make sure you use jars that can withstand boiling water; if you are unsure, a metal knife in the jar should eliminate the risk of a shattering jar.
Another option would be to boil the samples as a class so that students can see the preparation process and understand that the samples are being heated and then sealed to eliminate air movement while they are being cooled.
Engage: Ask: What do all living things need in order to survive? Students should eventually answer: oxygen. Ask: Do aquatic animals need oxygen? How do they get it? Students should be able to think of ways for oxygen to enter into the water: diffusion and plants. Hold up a glass of ice cubes and water and a glass of hot (if possible, steaming) water. Ask: What do you think could live in each of these glasses of water? Allow the students to make some guesses-don’t give them answers. Ask: What is necessary for living things to survive in water? They should be able to remember the lab they did testing for dissolved oxygen, and hopefully respond with “oxygen”. Ask: Do you think there is oxygen in very hot or very cold water? Allow students the time to write down the answers to these predictions.
Explore: Students will have access to all four water samples. They should test each sample two or three times, depending on the length of time you have in class. A good way to save time is to use the CHEMetrics test kits instead of the kits from Lamotte or HACH. Remind the students that even though all of the water temperatures are ‘cold’ or ‘cool’, two samples were prepared and ‘fixed’ at hot temperatures. Make sure that there are clear labels on the samples themselves or on the table where the samples are located. Do not open the sealed jars until the students are ready to test the water. They should create a chart which shows the temperatures at which the jars were sealed or tested – 5, 25, 50, and 100 ºC, and their results for each test.
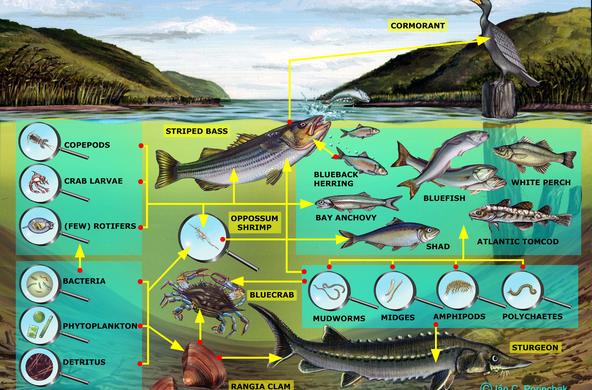
Explain: Cold water holds more dissolved oxygen than hot water, because as the temperature increases, the water releases some of the oxygen. The higher the temperature, the less dissolved oxygen. Ask: what do you think happens to the levels of dissolved oxygen in a river in the summer? At night? Students should be able to formulate answers to these questions based on their research. They should also be able to explain the importance of dissolved oxygen and the implications of low dissolved oxygen on aquatic ecosystems.
Extend: A variation on this experiment, if you don’t have time to complete the entire lab, is to take two bottles of soda, one that has been left in the sun or under a lamp, and another that has been left in a refrigerator for about an hour. Ask the students to predict the difference in the amount of bubbles that each bottle of soda will produce. When the bottles have reached different temperatures (you should be able to feel the difference), open them and observe. You should see more bubbles (carbon dioxide leaving the liquid) being created with the warm bottle. Even though this experiment involves the carbon dioxide, it is still the same principle as the solubility of oxygen in water. You can also do this with cups of the soda, and place one in the fridge. The cup in the fridge should ‘hold onto’ its bubbles longer because the carbon dioxide won’t dissolve out so quickly. Students could even do a taste test to see which one is ‘fizzier’.
Evaluate: Students will complete the lab report. Students should create a line graph of the class results using Excel to show the result for each temperature. Ask them to make predictions about what the dissolved oxygen concentration would be at different temperatures. Then, have them graph the seasonal data from the Hudson. This would be a good time to revisit the creation of trendlines, and their use in analyzing data.
Resources
Lesson Files
pdf
Worksheet with data embedded
pdf
Worksheet to use with Excel dataset
pdf
Worksheet with graphs embedded
Standards
Benchmarks for Science Literacy
5A Diversity of Life, 5D Interdependence of Life, 5E Flow of Matter and Energy, 9B Symbolic Representation, 9D Uncertainty, 12B Computation and Estimation, 12D Communication Skills, 12E Critical-Response SkillsNYS Standards
MST 1 - Mathematical analysis, scientific inquiry, and engineering design, MST 2- Informational Systems/ Information Technology, MST 4- Physical setting, living environment and nature of science, MST 5- Engineering and computer technology to satisfy societal needs, MST 6- Interconnectedness of mathematics, science, and technology (modeling, systems, scale, change, equilibrium, optimization), MST 7- Problem solving using mathematics, science, and technology (working effectively, process and analyze information, presenting results)Credits
Modified with permission from: “When the oxygen goes…” 1997. Living in Water, National Aquarium in Baltimore, Kendall Hunt Publishing, Iowa.