Objectives
Students will know the importance of soil as a water filter, and be able to discuss how the composition of the soil impacts its ability to filter pollutants.
Overview
Rating:
- Students discuss the importance of soil
- Students conduct activity to determine the composition of several soils
- They then test the ability of each soil to filter ‘pollutants’ and compare results
- Students have a competition to create the ‘best’ soil filter
Materials
- Instruct students to bring a baggie of soil from home – can be from their backyard, sediment from a nearby stream, whatever they want. This is the ‘unknown soil’
- pollutant mix ingredients: red + blue food coloring in water, vinegar, raisins, and dirt
- additional soil components for competition: baking soda, salt, and gravel
- optional: provide additional construction materials to incorporate more of a physical engineering component – toothpicks, mesh, plastic bottles, etc. You can also provide a table with all of these materials that any group can use if they wish.
Per group:
- 8-10 Paper cups of 2 sizes (or plastic bottles or coffee cans with holes punched in the bottom – see Procedure Part 2) – 4 large/4 small for Procedure Part 2; 1 large/1 small for Procedure Part 3
- 3 soil types: silty soil, clay soil, & sand—These provided soils are the ‘known’ soils.
- toothpicks (to place between large and small cups)
- graduated cylinder or other tool to measure liquid
- food coloring
- stopwatch
- pH test kit
- colored pencils (especially shades of blue, red, & purple)
Procedure
Preparation: Create ‘pollutant mix’ by adding raisins (e.g. solid organic waste), vinegar (a weak acid), and enough blue and red (or blue and yellow) food coloring to a gallon of water to turn it a deep purple (or green).
Optional: 3-10 days before completing this lesson, characterize the three known soils using the characterization protocol found on page 3 of the ‘Estimating Soil Texture’ handout. This procedure will provide great visuals and more robust quantification of soil components.
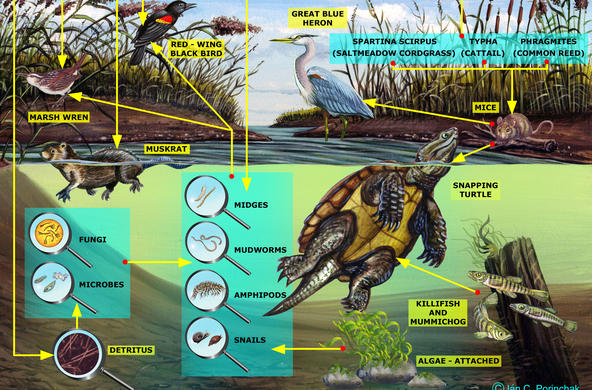
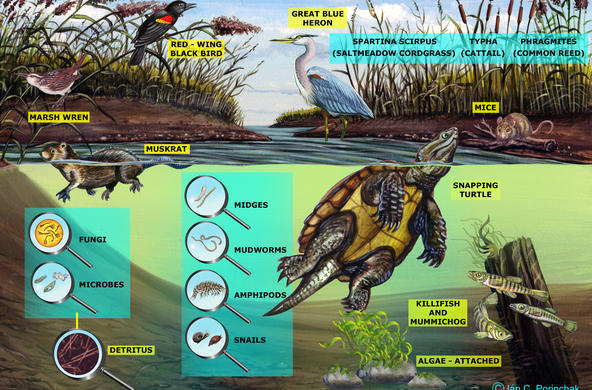
Engage: Hold up a cup of soil, and ask students to work in pairs to list the reasons why soil is important. Most students will not think about soil, or permeable surfaces, as important to water quality. Encourage students to think about the connections between land use and water, especially flooding.
Explore: Organize your students into groups of 3-4 students/ group. Distribute the three known soils, cups, etc. to each group.
Part 1: Characterizing the soils
Students will use the ‘Identifying Soil Texture by Feel’ protocol to determine the type of soil that they have. (See pages 214-4 and 215-5 of the ‘Estimating Soil Texture’) Have each student conduct the ‘feel’ tests and follow the flow chart for one of the known soils. Allow enough time for the students to switch soils, so that they have a good idea of what the textures of each of them look and feel like. Instruct them to follow the same procedure to find out the type of soil they brought from home.
Part 2: Testing ‘pollutants
Instruct each group in your method of choice for creating a filter.
Three easy ways to create a ‘filter’ are outlined below. Give the students whatever materials you find most accessible.
- Use paper cups: first cup is larger than the second. Punch holes in the bottom of the larger cup, and place inside the second, smaller cup. Slide a paper clip or toothpick along the side, between the larger and smaller cup to create a small space between the two cups; this will ensure that air can escape and a vacuum seal doesn’t form. The sand/soil/clay mixture can be added to the cup with the holes in the bottom.
- Use plastic bottles: cut the bottom off of a regular water bottle, and invert the bottle. Place a cotton ball over the neck of the bottle, and add the sand/soil/clay mixture above the cotton ball. The plastic bottle can be suspended inside of another plastic bottle from which the top has been cut off.
- Use coffee cans: Punch holes in the bottom of metal coffee cans, and line the bottom with mesh screen. The cans are filled with the soil sample and then are held above a graduated cylinder to capture the water.
Have each group do the following:
- Fill the top of each larger cup with 1.5 inches of one of each of the three known soils
- Add 1/4 cup of the ‘pollutant mix’ to each of the cups with soil in it.
- Compare the color and pH of the ‘pollutant’ before and after it has gone through each of the different kinds of soil. They should write their results on the ‘Student Worksheet’
- Make a prediction about how well the unknown soil will filter out the ‘pollutant mix,’ based on their observations of the other three soils and what they determined about the unknown soil’s composition.
Part 3: Students will design their own water filters, based on their observations in Parts 1 and 2.
Provide each group of students with the same materials: Each of the three known soil types, with the addition of: salt, baking soda, and gravel. Tell them that after the last storm, the city realized that some antiquated pipe systems were dumping storm runoff directly into the Hudson River.
This runoff came directly from the city streets and includes anything found there—oil, trash, chemicals, etc. In order to protect the health of the Hudson, the city is interested in installing rain gardens to help filter the runoff before it goes into the river. The students’ task is to design the optimal soil filtration system. The soil mixture that filters the runoff water the best will be used with appropriate wetland plants in the rain gardens. Tell them that you collected water from one of these pipes and will use it to test their filtration systems.
Each group should make their own mixture, being sure to write down the proportions of each soil type used (either measured with small cups or weighed in grams). Note that the students can also layer the soil types, or combine mixtures and layers in any way they see fit so long as they write down what they did.
Encourage students to create different types of soil mixtures—however, with only dirt used, it will be difficult to tell the color of the filtered water due to the soil particles. Allow the students ~20 minutes to create their filters. With the whole class watching, test each groups’ filter with your pre-made ‘pollutant mix.’ Be sure to pour the same amount of pollutant mix into each groups’ filter and to save the filtrates for comparison with the other groups. Test the pH of each group’s filtrate and write it on the cup. Have the class vote on the winner, based on final color and pH of the filtrate.
Explain: Soil acts as a natural filter for many substances found in water, including particulates, toxins, acids, and even some bacteria. Complex, layered soil mixtures slow down water runoff, trap larger particles, and buffer acidic soils. Clay soils (the smallest particles) have a negative charge and trap the positively charged blue food coloring. Amending soils with baking soda and some salts can increase the buffering capacity of soils. However, soil can also become contaminated by human activities, which then reduces the soil’s ability to act as a filter. Remember, even if water "looks" clean, it might not be!
Extend: Students could research the different ways that we filter water at our water treatment plants. Most treatment plants use some form of sand and coal to clean the water.
Evaluate: Collect students’ worksheets; for an exit ticket, ask students to explain how they would decide if a particular water sample is "dirty", and what methods might be useful for "cleaning" a water source.
Resources
Lesson Files
pdf
Lesson Worksheet
pdf
Estimating Soil Texture
Standards
Benchmarks for Science Literacy
2A Patterns and Relationships, 2B Mathematics, Science and Technology, 3B Design and Systems, 3C Issues in Technology, 4B The Earth, 4C Processes that shape the earth, 11C Constancy and Change, 12C Manipulation and Observation, 12D Communication SkillsNYS Standards
MST 1 - Mathematical analysis, scientific inquiry, and engineering design, MST 4- Physical setting, living environment and nature of science, MST 5- Engineering and computer technology to satisfy societal needs, MST 6- Interconnectedness of mathematics, science, and technology (modeling, systems, scale, change, equilibrium, optimization), MST 7- Problem solving using mathematics, science, and technology (working effectively, process and analyze information, presenting results)Credits
‘Estimating Soil Texture’ was created by Whiting, D., et al. Colorado State University Extension & USDA-ARS and is freely available online at:
https://culter.colorado.edu/~kittel/SoilChar(&RibbonTest)_handout.pdf&n…; (Colorado Master Gardener Estimating soil texture)