Objectives
Students will know where nitrogen exists and in which forms, and will be able to draw a diagram showing the movement of nitrogen in ecosystems.
Overview
Rating:
- Students answer formative assessment questions about nitrogen to illicit prior knowledge.
- Students complete the nitrogen cycle ‘game’ and discuss the different pathways and forms nitrogen can take in the environment.
- Students take notes and/or complete the Nitrogen Cycle Webquest.
- Optional: Students conduct investigations on nitrogen additions to a microcosm.
Materials
For the game:
- Laminated Nitrogen Cycle Game Descriptions
- Copies of the ‘Nitrogen Game Worksheet’
- One die for each station
- Nitrogen powerpoint
- Laminated pictures for each station (optional)
- Copies of the worksheet Nitrogen Cycle Webquest (optional)
- Nitrogen reading & questions (optional)
Procedure
Engage: As a formative assessment prompt, ask students whether nitrogen exists in:
- People
- Plants
- Animals
- Water
- Soil
- Air
This will help you evaluate whether students understand where nitrogen exists, and that it is an element that moves throughout the globe in different forms (organic, inorganic). You can use the prepared slides in the powerpoint.
Explore: Briefly explain the nitrogen ‘game’ students will be playing by pointing out the location of the different stations and then handing out the game sheet. Students will spend the next 20-25 minutes rotating through the stations (play the game at least 8 rounds) to gain an understanding of where nitrogen exists, and which forms it takes. You do not have to have students complete the “form of nitrogen” column. An extension idea is to have students explain the process that occurred to move nitrogen – did dentitrification happen? What about combustion? These are topics for higher level students that may be helpful as review. When students have completed the stations, ask them to summarize their journey.
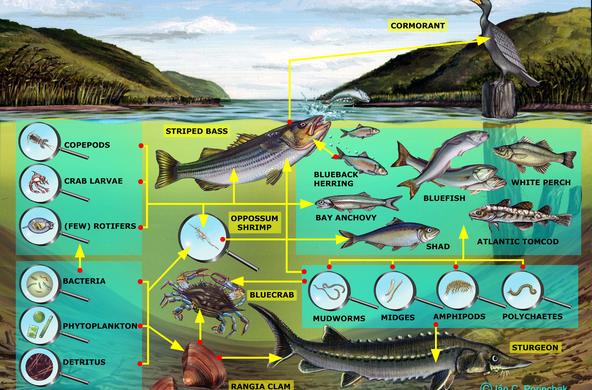
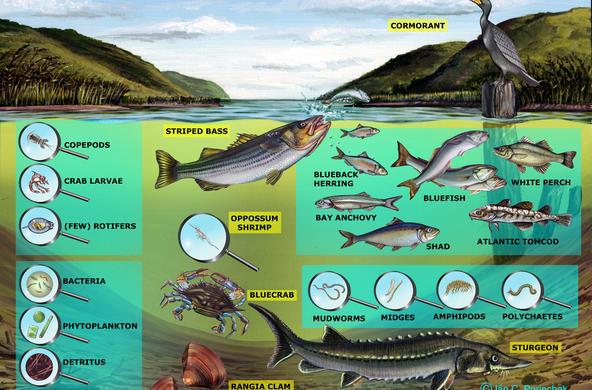
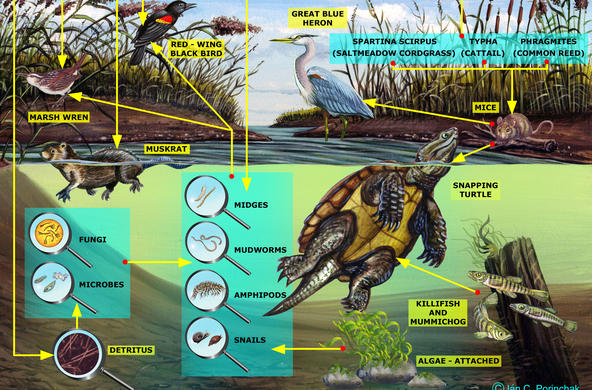
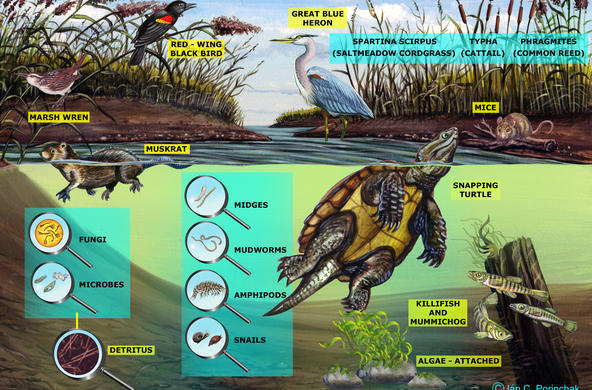
Explain: A powerpoint presentation is provided in order to help you with this topic; students may take notes. Nitrogen exists in many different forms. It is critical to organisms because it is part of proteins, chlorophyll, and genetic material (DNA, RNA, amino acids), and all organisms require nitrogen in order to live. Most exists as N2 gas in the atmosphere but plants can’t use it in this form. It has to be “fixed” or converted to a form plants can use, bonded to hydrogen or oxygen to form inorganic compounds such as ammonium (NH4) and nitrate (NO3). This can be done naturally, by lightning (transforming it into nitrate which then rains onto soil) or bacteria, or by humans, in the manufacture of fertilizer and the combustion of fossil fuels. Some of the bacteria live in the soil (releasing nitrogen during decomposition), and others live in the root nodules of certain kinds of plants. When plants take up nitrate, they use it to make proteins, amino acids, and nucleic acids after reducing it to nitrite and then ammonium. Animals receive the nitrogen they need for metabolism, reproduction, and growth by consuming living or dead organic matter. When an animal or plant dies, or an animal excretes, the nitrogen it releases is converted into ammonia (mineralization). Ammonia is then converted into nitrates (nitrification) for use by plants. The last step in the ‘cycle’ is denitrification, when nitrites are reduced back into nitrogen gas by bacteria.
One of the most amazing mutualistic symbiotic relationships involves nitrogen fixing bacteria and the roots of legumes (beans, peas, and clover). The bacteria live in the root nodules and receive sugar in exchange for fixing nitrogen for the plant. Thus, the relationship is beneficial to both organisms.
In the powerpoint: An example of a nitrogen atom’s pathway: An atom of N enters the watershed in an ammonium ion in rain, is converted to nitrate by bacteria in soil, washes out into the stream, is taken up by algae and incorporated into algae protein, washed downstream in dead plant, eaten by an oligochaete, eaten by a fish, passed out in feces, broken down into ammonium and then nitrate by bacteria, and then washed out to sea (use the powerpoint slide to accompany this).
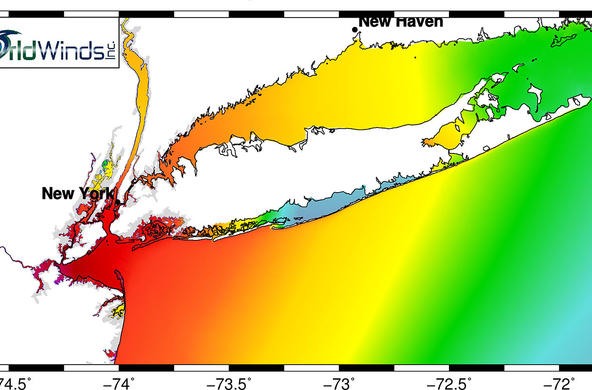
Within the last century, humans have more than doubled the amount of fixed nitrogen that is pumped into the atmosphere every year. In addition to acid rain, nitrogen pollution contributes to the formation of ground level ozone (which can cause breathing problems), acid rain, groundwater contamination, and eutrophication of coastal waters. Examples of a natural and a disturbed nitrogen cycle are provided as visuals in the powerpoint.
A teacher powerpoint is provided with additional nitrogen information as background.
Extend: Students should complete the ‘Nitrogen Cycle Webquest’, which explores issues related to excess nitrogen. Students may also want to read the background information on Nitrogen (see links below for reading & questions). For an experimental exploration of nitrogen, see the lesson “Nitrates & Marshes”.
Optional: Use the two lab worksheets (Lab 1 is more open-inquiry than Lab 2) to guide students through an investigation of how adding nitrogen affects a variety of ecological processes.
Evaluate: Students should complete the discussion questions of the worksheet. Focus on students’ answers to how people have changed the nitrogen cycle.
Resources
Lesson Files
pdf
Nitrogen Cycle Lab Version 1
pdf
Nitrogen Cycle Lab Version 2
pdf
Nitrogen Game Station Descriptions
pdf
Nitrogen in a disturbed watershed
pdf
Nitrogen in an undisturbed watershed
pdf
Nitrogen in Northeastern Watersheds
pdf
Nitrogen Powerpoint - Teacher Background
pdf
Nitrogen Game Stations
pdf
Nitrogen PowerPoint - for Classroom Use
pdf
Homework: Nitrogen Cycle Webquest
pdf
Nitrogen Cycle Game Worksheet
pdf
Nitrogen Cycle Game Worksheet Answer Key
Standards
Benchmarks for Science Literacy
5A Diversity of Life, 5D Interdependence of Life, 5E Flow of Matter and Energy, 9B Symbolic Representation, 9D Uncertainty, 12B Computation and Estimation, 12D Communication Skills, 12E Critical-Response SkillsNYS Standards
MST 1 - Mathematical analysis, scientific inquiry, and engineering design, MST 2- Informational Systems/ Information Technology, MST 4- Physical setting, living environment and nature of science, MST 5- Engineering and computer technology to satisfy societal needs, MST 6- Interconnectedness of mathematics, science, and technology (modeling, systems, scale, change, equilibrium, optimization), MST 7- Problem solving using mathematics, science, and technology (working effectively, process and analyze information, presenting results)Credits
Bennett, V. 2002. “Eutrophication-A Project Lab for Multi-Section Lab Courses.” Tested studies for laboratory teaching, Vol 23, M.A. O’Donnell, editor. Proceedings of the 23rd Workshop Conference of the Association for Biology Laboratory Education (ABLE), 392 pages. Ecological Society of America.