From the 2019 Cary report Road Salt: The Problem, The Solution, and How to Get There
There is a legacy effect of salt in the environment, which means that concentrations in surface and groundwater will increase, perhaps for decades, even if we stop using road salt today. So, the average concentration of 48 mg/L we see today could be much higher in the future.
Road salt can also damage metal and concrete, contaminate drinking water, damage roadside vegetation, and accumulate in streams, lakes, reservoirs, and groundwater harming aquatic plants and animals.
Trends show that, even in relatively rural areas, road salt is accumulating in our waterways. Because it can take decades for road salt to flush out of a watershed, increases in concentrations of salt may be seen even after its use has stopped. The combination of alarming increases in salt together with the time required for increases to cease indicate that it’s important to address the problem now.
While safe roads are of utmost importance, recent research indicates that we can achieve safety while being more efficient and careful with our road salt. By combining efforts to improve efficiency in road salt use with alternative chemicals in targeted areas, we can make a difference and improve conditions for ourselves and future generations.
Application tips & best practices
Adding too much salt to an icy surface is a waste of money and can only increase damage to concrete, metal, drinking water, and vegetation. It is a good rule of thumb to use deicers sparingly. Deciding how much to use depends on the deicer. A successful rate for rock salt is about a handful per square yard.
If using calcium chloride, the amount needed is less—about a handful for every 3 square yards. Here are some precautionary steps you can take to decrease the amount of deicer you’ll need.
- Shovel the snow early and often. If the temperature drops after a snowstorm, the snow can turn icy and be harder to remove
- The more scraping and removal of ice that you can do, the less deicer you will need to use. Deicers work best on a thin layer of ice
- After you remove all of the snow and ice, sprinkle salt sparingly
- As the sun comes out or the temperature rises, the deicer will make a slushy mixture of water and ice. Remove this before the temperature drops again and you should have an ice-free surface until the next storm.
If you are a municipality, highway department, or part of a winter maintenance staff, we also offer tips and best practices for reducing road salt application.
Alternative deicers
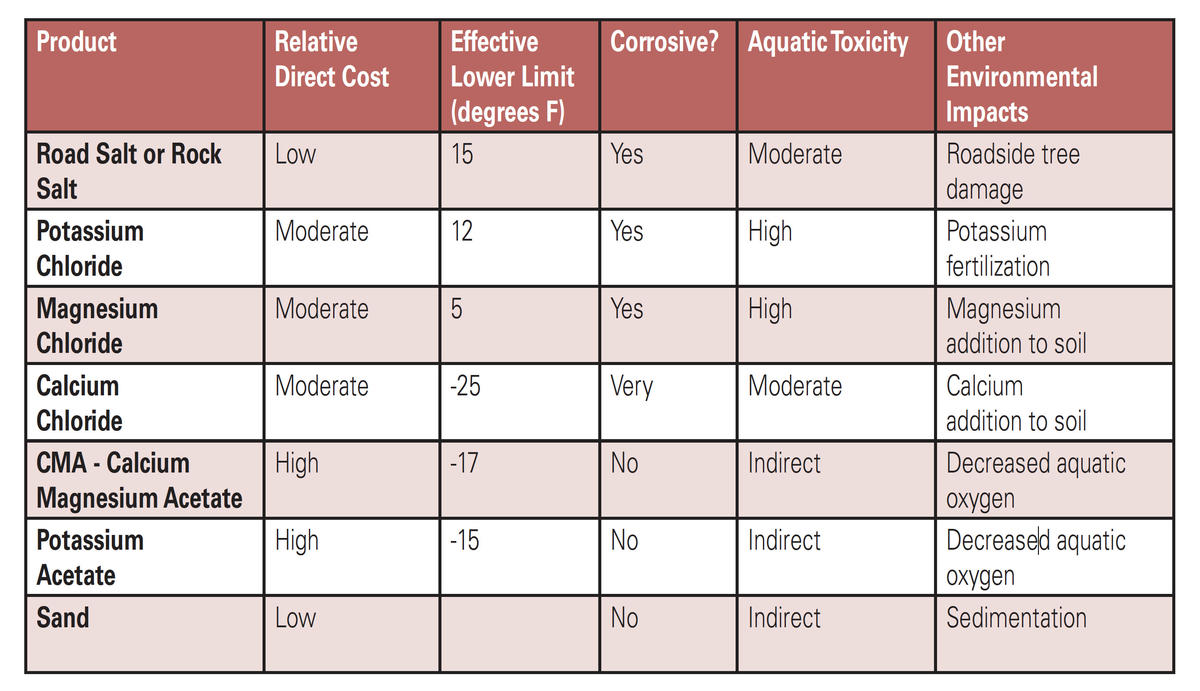
Road salt alternatives
Alternative salt-based deicers have changed very little in recent years. Interest in agro-based alternatives has expanded greatly although salt is the active ingredient in most of these products. Products include:
- Corn steepwater (a byproduct of wet corn milling)
- Cheese and pickle brine
- Fermentation byproducts (i.e., from beer and wine waste)
- De-sugared molasses (a byproduct of sugar beet processing)
Agro-products can lower the freezing point of chloride-based salts and increase the amount of time salts remain on pavement. They can also improve sunlight absorption and reduce the corrosivity of salt solutions. When washed into streams and lakes, many agro-based deicers have an impact, as would be expected when any organic material (such as wastewater) is released into a water body.
High levels of organic material can deplete oxygen, stimulate algae, and cause fishkills. These agro-based deicers also tend to be more expensive. Other alternatives to rock salt include different types of salt or sand, which vary in effectiveness and impact








