Objectives
Students will know the factors that change dissolved oxygen levels and be able to design an experiment to test their ideas.
Overview
Rating:
1. Students discuss the possible implications of low dissolved oxygen levels in aquatic environments.
2. In groups, students hypothesize different ways to change the DO.
3. Students create a research plan for approval by the teacher.
4. Students carry out their experiment, and summarize their results for the class.
Materials
- Mason jars
- Hot plates
- Dissolved oxygen kits
- Aquatic plants
- Snails
- Salt
- Thermometer
- BTB
- Fish tank bubbler
- Nitrogen fertilizer
- Ice
- Foil
- NaOH solution (optional)
Procedure
Preparation: This is an advanced lab that requires students to be good at designing their own experiments, following directions, collecting data, and making conclusions from their observations. This should not be the first inquiry-based lab that the students do during the year, because they will need experience on how to complete experiments on their own. If you would like to scaffold the lab, you can do the first part as a class and then allow the students to work in groups for the second part.
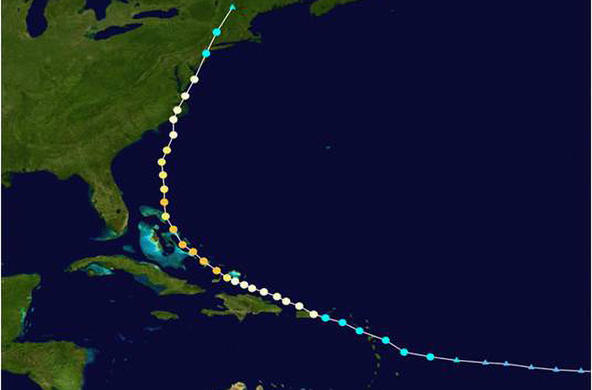
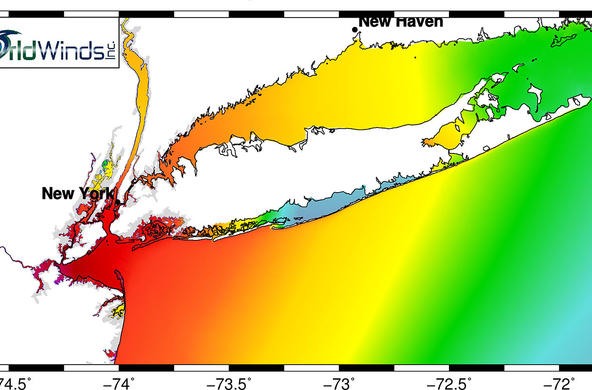
Engage: Show students an aerial image of low dissolved oxygen areas in Long Island Sound, which can be found at the end of this lesson. Ask: What might cause this condition in the Sound? Discuss the possible implications of low levels of dissolved oxygen in aquatic ecosystems. Ask: Why is dissolved oxygen important? What are the ways that oxygen gets into the water? What types of things might affect the dissolved oxygen levels? Discuss the terms hypoxic and anoxic.
Give students a vial or small glass of water, and add a small amount of BTB (this can be done in groups or as a demonstration). Using a straw, ask the student(s) to blow into the tube or glass. If using a vial, squeeze a cotton ball into the top to avoid the solution squirting out. The color of the BTB will turn yellow, indicating the presence of carbon dioxide. Additing a few drops of 0.01m NaOH solution will turn it back to blue, removing the carbon dioxide.
Explore: In groups, students should discuss the possible affects of different variables on dissolved oxygen levels. Depending on the ability of your class, you may want to help define these variables:
- Temperature: Students heat and cool water, testing the DO levels at each temperature. Use caution with boiling water-sealing the water in a jar and allowing it to cool to room temperature before testing is important. A good protocol for heating water can be found in the lesson plan “Dissolved Oxygen and Temperature” if students are stuck.
- Presence of aquatic plants: Using BTB or dissolved oxygen test kits, students can measure the levels of DO with and without aquatic (submerged) plants. This usually takes at least 24 hours to see a response. Make sure that students think about the impacts of respiration as well. Using light as an additional variable will help students think about photosynthesis and respiration. If using BTB, students can turn the solution yellow before starting the experiment by carefully blowing bubbles into solution. Adding the plants to the yellow solution will turn it back to blue, indicating the presence of oxygen. This will not, however, give students an idea of the amount of oxygen in the water, just the presence or absence of oxygen.
- Types of aquatic plants: Set up three tanks: a control without plants, one with floating plants, and one with submerged plants. The tanks/jars need to be left for one-two weeks, undisturbed, in order to see a response in the DO levels. If you are using a HACH kit, it is best to use a larger aquarium than a mason jar, because you will have to drop the sample bottle into the water. If you are using Chemetrics test kits or a probe, you can let the students use smaller containers for the water.
- Aquatic animals: Using BTB or DO test kits, students can set up jars with and without aquatic animals. This usually takes about 24 hours to see a response. The jars or tanks should be sealed. The BTB will turn yellow with the presence of carbon dioxide. Sample questions students could ask: Do snails take in or give off CO2? Do elodea plants take in or give off CO2? What about at night?
- Salinity: Adding salt to the water decreases the ability of oxygen to dissolved in the water. Allowing students to experiment with different concentrations of salt water is a quick way to look at DO changes.
- Aeration/surface exposure: The movement of water is one of the primary ways for oxygen to enter a water body. Students can set up two jars, one with, and one without, an aeration pump (from an aquarium). They can also simply cover a water sample for several days and measure the DO level in a covered vs an uncovered sample.
- Decomposition: Adding fertilizer to water (use pond water for this so that you have some algal cells) causes an increase in plant growth, which will reduce the amount of oxygen available as the plants die and are broken down by bacteria. Students can either add fertilizer or add dead leaves to a water sample. The fertilizer approach takes much longer, often two weeks or more. Dead leaves decompose quickly, and covering the jar or tank with plastic wrap increases the decomposition rate.
Note: Altitude and tidal cycles also affect dissolved oxygen levels but is not easily simulated in the classroom. However, some students have set up experiments in which they simply introduce ‘new’ water every six hours to simulate the tides. Students should design an experiment which tests the impact of one variable on dissolved oxygen levels. Depending on the amount of time you have for this activity, students could do a preliminary experiment and then revise their test protocol and repeat it. When advising students on the type of experiment to pursue, be conscious of the amount of time you want to spend on the experiment.
Explain: Dissolved oxygen is one of the most important components of water quality, and is often the limiting factor in a particular environment. Dissolved oxygen levels in the Hudson fluctuate with changes in the tides, aquatic vegetation, and salinity. Since the Hudson has both tides and a current, it is not usually oxygen limited except for in the water chestnut beds that grow along the freshwater shoreline. However, there are significant problems with thermal pollution and nitrogen additions. See the graphic titled ‘Nitrogen Inputs to the Hudson’ at the end of this lesson, and the photo of thermal pollution from Indian Point. Another interesting discussion revolves around the definition of ‘hypoxic’. Although everyone agrees that anoxic means no dissolved oxygen, there is significant variation among various agencies as to what constitutes hypoxic waters. According to a New York Times article published on Nov. 11, 2007, Connecticut has reduced the amount of nitrogen being released into the LIS since 2002 by allowing the sewage-treatment plants to trade pollution credits, but there is still a significant incidence of hypoxia every summer. Under current CT Department of Environmental Protection policy, hypoxia is defined as water that is below 3.5 mg/L. This was changed from 3.0 mg/L in 2004. To illustrate what this means, it is helpful to talk about the 2005 summer hypoxia survey the CT DEP completed. Under the new DO standard of 3.5 mg/L of dissolved oxygen, the maximum area affected by hypoxia, seen during the August 2 – 4, 2005 survey, was 300 square miles. Under the old DO standard of 3.0 mg/L of dissolved oxygen, the maximum area affected by hypoxia was 177 square miles. See the website for details about other years: http://www.ct.gov/dep/site/default.asp . Studies in the Hudson, however, define hypoxia as below 2.5 mg/L. The EPA defines hypoxic as below 2-3 mg/L.
Extend: Students should research the implications of hypoxic waters on aquatic organisms to answer the following question: Should there be a nationwide standard of hypoxia? Why or why not?
Evaluate: Students should present the results of their work to the class, and submit a lab report using the accompanying worksheet as a guideline.
Resources
Lesson Files
pdf
Student worksheet