Objectives
Students will know at what level of salt concentration aquatic organisms are affected, and be able to explain the results of an experiment to determine these levels.
Overview
Rating:
- Students will set up microcosms to test toxicity of different levels of salt pollution on water fleas or duckweed
- Students record results for 2-5 days
- Students discuss the implications of salt pollution based on their data
Materials
For Daphnia experiment:
- 5-10 organisms per container
- Deionized rock salt
- Distilled water
- 14 beakers or cups
- Pipette
For duckweed experiment:
- 3 plants per container
- Deionized rock salt
- Distilled water
- 14 beakers or cups
- Paper filter for petri dish
- Tweezers
Procedure
Preparation: You should prepare 6 different stock solutions, or variations on the concentrations below. This can be done either with the students in the lab or ahead of time.
- Stock solution of 9.5 grams salt for 500 mL of water = 19,000 mg/L
- Stock solution of 2 grams salt for 500 mL of water = 4000 mg/L
- Stock solution of 1 gram salt for 500 mL of water = 2000 mg/L
- Stock solution of 1 gram salt for 1000 ml of water = 1000 mg/L
- Stock solution of 0.5 gram salt for 1000 ml of water = 500 mg/L
- Stock solution of 0.1 gram salt for 1000 ml of water, then take 500 ml of this water and add another 500 mL of regular water = 50 mg/L
The average concentration for the Wappinger Creek has been around 50 mg/L for the last few years. Ocean water generally has about 19,000 mg/L. The salt front of the Hudson is wherever the concentration reaches 100 mg/L. In the estuarine parts of the river, salinity ranges from 500 mg/L to 3500 mg/L and higher.
Purchase a Daphnia culture kit at least 2 weeks before you plan to do the experiment, to ensure that you have a healthy culture. You should try and use newly hatched Daphnia in order to minimize the differences in the sizes of the animals. Because the appearance of resting eggs indicates a poor culture environment, do not use Daphnia with resting eggs. Students can do these next steps to minimize the preparation time.
Twenty-four hours before beginning the experiment, remove all the females bearing embryos from the stock culture and place them in 400-mL beakers containing 300 mL of spring or stream water and the appropriate amount of food. Five beakers, each containing 10 adults, usually will supply enough young individuals for one toxicity test.
When you are ready to begin your bioassay, choose young (small) Daphnia from these cultures. Introduce the same number of neonates (at least 10) into each test vessel and control using a plastic, disposable pipette with a 5-mm diameter. Be sure to release the young below the surface to avoid killing them by trapping air under their carapaces. Record the time and number of young introduced into each labeled vessel.
Duckweed can be ordered from scientific supply companies, or collected from area ponds. If you are using duckweed, you will need enough for each container to have three plants. Separate them in petri dishes; it is easier if the petri dishes have coffee filters in them, so that the plants can be easily seen and moved with tweezers.
Engage: Have two glasses ready at the front of the room, one with a tablespoon of salt dissolved in it, and another without any salt. Ask for a volunteer to come up and taste the two water samples; make sure students know they can spit out the samples if they want to. Have them explain to the class what they tasted, and ask students if this is a form of pollution or not. Ask students to think about the winter application of road salt, and what happens to this salt after it is left on the road. Are there other sources of salt to the watershed that students can think of? Make a list on the board; if it is incomplete, students can add to it throughout the class.
Explore: Students will receive the materials and begin working on the lab, following the directions on the lab sheet. You will need to leave the Daphnia in the salt solution overnight in order to see a response (4-6 hours is usually enough). It is recommended that you allow the Daphnia to remain in the solutions for 48 hours. The duckweed need to be left for at least five days in order to see a response.
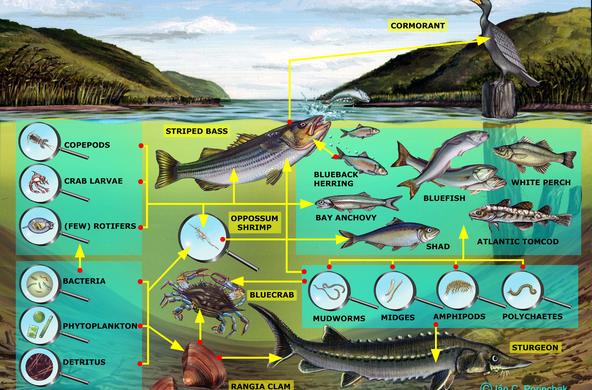
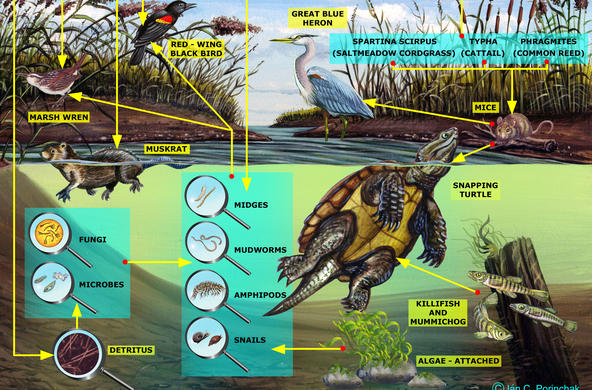
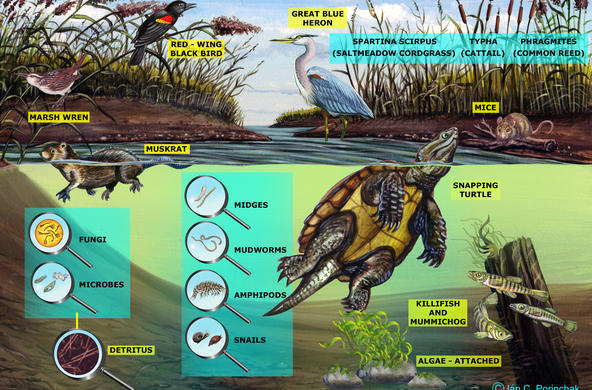
Explain: Besides road salt, there are other sources of salt to our watersheds. Water softeners, runoff from sewage systems, deposition, and rock weathering are some other ways that salt can enter the ecosystem. Students will explore these other sources of salt pollution in the following lesson. When road salt is applied, it eventually dissolves into water and moves through the soil to groundwater or into surface water. Salt affects organisms even below lethal concentrations, by altering the drifting of aquatic insects (drifting is a form of migration and movement). Another problem with creating consistently salty, unnatural conditions is the resulting ability of salt-tolerant invasive species to move into an area. Generally, zooplankton die at a concentration of 1000 mg/L, although students should note that salt accumulates over time, and the longer the organisms are in a high salt environment, the more toxic the effects.
Extend: Students could calculate the Lethal Concentration 50, or LC50, which is used by the EPA to determine the toxicity of certain pollutants. When 50% of the organisms die, that concentration is considered the upper limit. For this experiment, consider that when 7 out of 10 Daphnia die at a specific concentration, that is recorded as 100% mortality. Once students have determined the mortality threshold, they could design a second experiment to determine the specific level at which Daphnia are no longer successful.
Evaluate: Students will submit their lab reports.
Resources
Lesson Files
pdf
Lab sheet
Standards
Benchmarks for Science Literacy
1B Scientific Inquiry, 2B Mathematics, Science and Technology, 5A Diversity of Life, 5D Interdependence of Life, 11C Constancy and Change, 12C Manipulation and Observation, 12D Communication SkillsNYS Standards
MST 1 - Mathematical analysis, scientific inquiry, and engineering design, MST 3- Mathematics in real-world settings, MST 4- Physical setting, living environment and nature of science, MST 6- Interconnectedness of mathematics, science, and technology (modeling, systems, scale, change, equilibrium, optimization), MST 7- Problem solving using mathematics, science, and technology (working effectively, process and analyze information, presenting results)Credits
Experiment adapted from: Environmental Inquiry, Cornell University and Penn State University, 2006. http://ei.cornell.edu/